HPLC pigments along the US GEOTRACES East Pacific Zonal Transect from the R/V Thomas G. Thompson TN303 cruise in the tropical Pacific from Peru to Tahiti during 2013 (U.S. GEOTRACES EPZT project)
Project
Program
| Contributors | Affiliation | Role |
|---|---|---|
| Moffett, James W. | University of Southern California (USC-HIMS) | Lead Principal Investigator, Contact |
| Cutter, Gregory A. | Old Dominion University (ODU) | Co-Principal Investigator |
| German, Christopher R. | Woods Hole Oceanographic Institution (WHOI) | Co-Principal Investigator |
| Bidigare, Robert R. | University of Hawaiʻi at Mānoa (SOEST) | Analyst |
| Gegg, Stephen R. | Woods Hole Oceanographic Institution (WHOI BCO-DMO) | BCO-DMO Data Manager |
Bidigare Pigments
Notes on the data:
Parameter names were modified on 16 May 2017 to comply with SCOR Working Group 78 (http://www.scor-int.org/SCOR_WGs_WG78.htm).
Pigment values are concentrations in ng/L.
Where the value is shown as zero, this could more properly be stated as "less than limit of detection".
See LoD estimates below.
Filtered volume amounts were obtained from the hand written cast sheets.
They are listed with these data simply for reference in case one has different values listed elsewhere.
Station 21: No depths are listed for Station 21 because these were not available on the cast sheets.
The response factor for DIADINO was used to calculate DIATO values.
Station 2: It is possible that some HEX FUCO is present. The high levels of FUCO at this station resulted in correspondingly large cis-fuco peaks, one of which eluted at the same retention time as HEX FUCO would elute if present, so HEX FUCO cannot be quantified. The size of the peak is about "right" for it to be due mostly or entirely to cis-fuco, but some contribution from HEX FUCO cannot be ruled out.
Station 4, sample 2373: Values for this one are oddly high for its location in the profile. I see no obvious errors on my end. Possible filter in wrong cryotube?
LoD Estimates:
Volume Filtered (L) 1.84 2.00 3.80 4.00
LoD for chlorophylls (ng/L) 4.8 4.4 2.3 2.2
LoD for carotenoids (ng/L) 3.2 2.9 1.5 1.5
These values are calculated based on LoD for CHL A of 0.59 ng/injection and for ZEA of 0.39 ng/injection, with an injection volume of 0.2 mL and an extract volume of 3.0 mL.
Pigment values <LOD were removed from the data set and reported as zeroes. Zeroes for this data set should be considered to mean <LOD.
Pigment = Abbreviation
Chlorophyllide a = CHLIDE_A
Chlorophyll C = CHL_C_TOT
Peridinin = PERID
19'-butanoyloxyfucoxanthin = BUT_FUCO
Fucoxanthin = FUCO
19'-hexanoyloxyfucoxanthin = HEX_FUCO
Prasinoxanthin = PRAS
Violaxanthin = VIOLA
Diadinoxanthin = DIADINO
Alloxanthin = ALLO
Diatoxanthin = DIATO
Lutein = LUT
Zeaxanthin = ZEA
Chlorophyll b = CHL_B
Alpha-carotene = ALPHA_CAR
Beta-carotene = BETA_CAR
Divinyl chlorphyll a = DV_CHL_A
Monovinyl chlorophyll a = CHL_A
Total chlorophyll a = CHL_A_DV_CHL_A
Sampling and Analytical Methodology: (also described in the Analysis of Pigments PDF)
Water samples were collected in opaque bottles and filtered onto 25-mm Whatman GF/F glass fiber filters (nominal porosity of 0.7 um) using vacuum filtration (7-10 in Hg). Sample filters were folded in half and individually stored in folded packets of heavy-duty aluminum foil, then placed immediately in liquid nitrogen.
Filters for pigment analyses were extracted in 3 mL of HPLC-grade acetone in culture tubes along with 50 uL of an internal standard (canthaxanthin) at 4C for 24 hours. The extracts were then brought to room temperature, vortexed, and centrifuged for 5 minutes to remove cellular and filter debris. Mixtures of 1-mL extract plus 0.3-mL HPLC grade water were prepared in opaque autosampler vials and 200 uL injected onto a Varian 9012 HPLC system equipped with a Varian 9300 autosampler, a Timberline column heater (26C), and a Waters Spherisorb 5-um ODS-2 analytical (4.6 x 250 mm) column and corresponding guard cartridge (7.5 x 4.6 mm). Pigments were detected with a ThermoSeparation Products UV2000 detector (l1 = 436, l2 = 450). A ternary solvent system was used for pigment analysis: Eluent A (methanol:0.5 M ammonium acetate, 80:20, v/v), Eluent B (acetonitrile:water, 87.5:12.5, v/v), and Eluent C (100 % ethyl acetate). Solvents A and B contained an additional 0.01 % 2,6-di-ter-butyl-p-cresol (0.01 % BHT, w/v; Sigma-Aldrich) to prevent the conversion of chlorophyll a into chlorophyll a allomers. The linear gradient used for pigment separation was a modified version of the Wright et al. (1991) method: 0.0’ (90 % A, 10 % B), 1.00’ (100 % B), 11.00’ (78 % B, 22 % C), 27.50’ (10 % B, 90 % C), 29.00’ (100 % B), 30.00’ (100 % B), 31.00’ (95 % A, 5 % B), 37.00’ (95 % A, 5 % B), and 38.00’ (90 % A, 10 % B) (Bidigare et al., 2005). Eluent flow rate was held constant at 1.0 mL per min.
Pigment peaks were identified by comparison of retention times with those of pure pigment standards (DHI Lab Products, Hørsholm, Denmark) and extracts prepared from phytoplankton reference cultures. Pigment concentrations were calculated using internal and external standards, and expressed as concentrations (ng pigment per L of filtered seawater). A dichromatic equation was used to resolve mixtures of monovinyl and divinyl chlorophyll a spectrally (Bidigare and Trees, 2000).
Stable detector response was confirmed on a weekly basis using freshly prepared chlorophyll a standard. Chlorophyll a standard injections were also run concurrently with samples as a daily QC check. An internal standard was used to account for extraction volume variability.
Lower limit of detection for the HPLC analytical method is 0.39 ng per inj for carotenoids and 0.59 ng per inj for chlorophylls. Precision for the analytical method is 0.81 %RSD for carotenoids and 1.40 %RSD for chlorophylls as determined from the mean (x̅) and standard deviation (s) of replicate injections (n = 30) and calculated as 100 times |s/x̅|. Accuracy for this analytical method is estimated as ~0.5% based on the uncertainty in the absorbance measurement of chlorophyll a standard.
References:
Bidigare, R.R., Van Heukelem, L., Trees, C.C., 2005. Analysis of algal pigments by high-performance liquid chromatography. In: Andersen, R. (Ed.), Algal Culturing Techniques. Academic Press, pp. 327-345.
Wright, S.W., Jeffrey, S.W., Mantoura, R.F.C., Llewellyn, C.A., Bjornland, T., Repeta, D., Welschmeyer, N., 1991. Improved HPLC method for the analysis of chlorophylls and carotenoids from marine phytoplankton. Marine Ecology Progress Series 77, 183-196.
Bidigare, R.R., Trees, C.C., 2000. HPLC phytoplankton pigments: sampling, laboratory methods, and quality assurance procedures. In: Mueller, J.L., Gargion, G. (Eds.), Ocean Optics Protocols for Satellite Ocean Color Sensor Validation, Revision 2, NASA Technical Memo, 2000209966, pp. 154-161.
For data processing, refer to the Analysis of Pigments PDF.
BCO-DMO Processing Notes
- Generated from original .xlsx file "GEOTRACES_May2014_final.xlsx, sheet: Pigment data" contributed by Jim Moffett
- Station, cast, bottle and sample metadata inserted from EPZT Master_Event sheet
- Parameter names were initially edited to conform to BCO-DMO naming convention found at Choosing Parameter Name
- Parameter names were again modified on 16 May 2017 to be compliant with SCOR Working Group 78
| File |
|---|
Bidigare_Pigments.csv (Comma Separated Values (.csv), 32.60 KB) MD5:e5a53bcfe2fe78c1e5e807abe1b53873 Primary data file for dataset ID 558538 |
| Parameter | Description | Units |
| EXPOCODE | expedition code assigned by the CCHDO: NODCShipCodeYearMonthDay | text |
| SECT_ID | cruise section identification number | text |
| STNNBR | station number | dimensionless |
| CASTNO | cast number | dimensionless |
| GEOTRC_EVENTNO | GEOTRACES Event Number | dimensionless |
| DATE | Station Date (GMT) | YYYYMMDD |
| TIME | Station Time (GMT) | HHMM |
| ISO_DATETIME | Date/Time (ISO formatted) (GMT) | YYYY-MM-DDTHH:MM:SS[.xx]Z |
| LATITUDE | Station Latitude (South is negative) | decimal degrees |
| LONGITUDE | Station Longitude (West is negative) | decimal degrees |
| GEOTRC_INSTR | GEOTRACES Instrument Id | text |
| GEOTRC_SAMPNO | GEOTRACES Sample Number | dimensionless |
| SAMPNO | sequential sample number within a cast | dimensionless |
| BTLNBR | Bottle Number | text |
| BTLNBR_FLAG_W | Bottle Quality Flag | dimensionless |
| BTL_DATE | Bottle Date (GMT) | YYYYMMDD |
| BTL_TIME | Bottle Time (GMT) | HHMM |
| BTL_ISO_DATETIME | Bottle Date/Time (ISO formatted) (GMT) | YYYY-MM-DDTHH:MM:SS[.xx]Z |
| BTL_LAT | Bottle Latitude (South is negative) | decimal degrees |
| BTL_LON | Bottle Longitude (West is negative) | decimal degrees |
| Station | Station Number | dimensionless |
| Depth | Sample Depth | meters |
| L_FILTERED | Liters filtered | liters |
| CHLIDE_A | Chlorophyllide a | ng/L |
| CHL_C_TOT | Chlorophyll c | ng/L |
| PERID | Peridinin | ng/L |
| BUT_FUCO | 19'-butanoyloxyfucoxanthin | ng/L |
| FUCO | Fucoxanthin | ng/L |
| HEX_FUCO | 19'-hexanoyloxyfucoxanthin | ng/L |
| PRAS | Prasinoxanthin | ng/L |
| VIOLA | Violaxanthin | ng/L |
| DIADINO | Diadinoxanthin | ng/L |
| ALLO | Alloxanthin | ng/L |
| DIATO | Diatoxanthin | ng/L |
| LUT | Lutein | ng/L |
| ZEA | Zeaxanthin | ng/L |
| CHL_B | Chlorophyll b | ng/L |
| ALPHA_CAR | Alpha-carotene | ng/L |
| BETA_CAR | Beta-carotene | ng/L |
| DV_CHL_A | Divinyl chlorphyll a | ng/L |
| CHL_A | Monovinyl chlorophyll a | ng/L |
| CHL_A_DV_CHL_A | Total chlorophyll a | ng/L |
| Dataset-specific Instrument Name | |
| Generic Instrument Name | CTD Sea-Bird SBE 911plus |
| Generic Instrument Description | The Sea-Bird SBE 911 plus is a type of CTD instrument package for continuous measurement of conductivity, temperature and pressure. The SBE 911 plus includes the SBE 9plus Underwater Unit and the SBE 11plus Deck Unit (for real-time readout using conductive wire) for deployment from a vessel. The combination of the SBE 9 plus and SBE 11 plus is called a SBE 911 plus. The SBE 9 plus uses Sea-Bird's standard modular temperature and conductivity sensors (SBE 3 plus and SBE 4). The SBE 9 plus CTD can be configured with up to eight auxiliary sensors to measure other parameters including dissolved oxygen, pH, turbidity, fluorescence, light (PAR), light transmission, etc.). more information from Sea-Bird Electronics |
| Dataset-specific Instrument Name | GO-FLO |
| Generic Instrument Name | GO-FLO Bottle |
| Dataset-specific Description | CTD bottle data from 30-ODF/SIOR (Ocean Data Facility 12 bottle, 30 liter Niskin rosette) |
| Generic Instrument Description | GO-FLO bottle cast used to collect water samples for pigment, nutrient, plankton, etc. The GO-FLO sampling bottle is specially designed to avoid sample contamination at the surface, internal spring contamination, loss of sample on deck (internal seals), and exchange of water from different depths. |
| Dataset-specific Instrument Name | Niskin bottle |
| Generic Instrument Name | Niskin bottle |
| Generic Instrument Description | A Niskin bottle (a next generation water sampler based on the Nansen bottle) is a cylindrical, non-metallic water collection device with stoppers at both ends. The bottles can be attached individually on a hydrowire or deployed in 12, 24, or 36 bottle Rosette systems mounted on a frame and combined with a CTD. Niskin bottles are used to collect discrete water samples for a range of measurements including pigments, nutrients, plankton, etc. |
TN303
| Website | |
| Platform | R/V Thomas G. Thompson |
| Report | |
| Start Date | 2013-10-25 |
| End Date | 2013-12-20 |
| Description | A zonal transect in the eastern tropical South Pacific (ETSP) from Peru to Tahiti as the second cruise of the U.S.GEOTRACES Program. This Pacific section includes a large area characterized by high rates of primary production and particle export in the eastern boundary associated with the Peru Upwelling, a large oxygen minimum zone that is a major global sink for fixed nitrogen, and a large hydrothermal plume arising from the East Pacific Rise. This particular section was selected as a result of open planning workshops in 2007 and 2008, with a final recommendation made by the U.S.GEOTRACES Steering Committee in 2009. It is the first part of a two-stage plan that will include a meridional section of the Pacific from Tahiti to Alaska as a subsequent expedition.
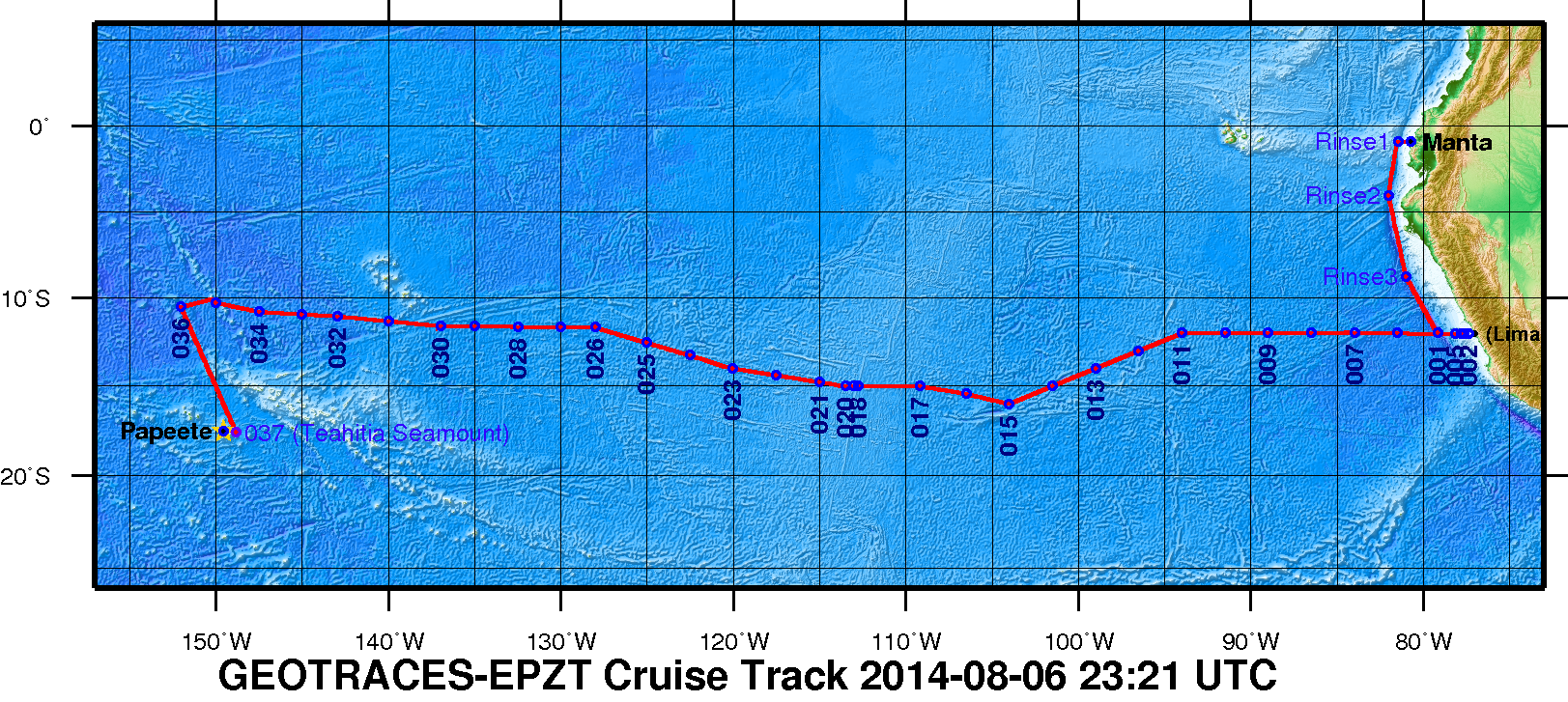
Figure 1. The 2013 GEOTRACES EPZT Cruise Track. [click on the image to view a larger version]
Additional cruise information is available from the Rolling Deck to Repository (R2R): http://www.rvdata.us/catalog/TN303 |
U.S. GEOTRACES East Pacific Zonal Transect (GP16) (U.S. GEOTRACES EPZT)
From the NSF Award Abstract
The mission of the International GEOTRACES Program (https://www.geotraces.org/), of which the U.S. chemical oceanography research community is a founding member, is "to identify processes and quantify fluxes that control the distributions of key trace elements and isotopes in the ocean, and to establish the sensitivity of these distributions to changing environmental conditions" (GEOTRACES Science Plan, 2006). In the United States, ocean chemists are currently in the process of organizing a zonal transect in the eastern tropical South Pacific (ETSP) from Peru to Tahiti as the second cruise of the U.S.GEOTRACES Program. This Pacific section includes a large area characterized by high rates of primary production and particle export in the eastern boundary associated with the Peru Upwelling, a large oxygen minimum zone that is a major global sink for fixed nitrogen, and a large hydrothermal plume arising from the East Pacific Rise. This particular section was selected as a result of open planning workshops in 2007 and 2008, with a final recommendation made by the U.S.GEOTRACES Steering Committee in 2009. It is the first part of a two-stage plan that will include a meridional section of the Pacific from Tahiti to Alaska as a subsequent expedition.
This award provides funding for management of the U.S.GEOTRACES Pacific campaign to a team of scientists from the University of Southern California, Old Dominion University, and the Woods Hole Oceanographic Institution. The three co-leaders will provide mission leadership, essential support services, and management structure for acquiring the trace elements and isotopes samples listed as core parameters in the International GEOTRACES Science Plan, plus hydrographic and nutrient data needed by participating investigators. With this support from NSF, the management team will (1) plan and coordinate the 52-day Pacific research cruise described above; (2) obtain representative samples for a wide variety of trace metals of interest using conventional CTD/rosette and GEOTRACES Sampling Systems; (3) acquire conventional JGOFS/WOCE-quality hydrographic data (CTD, transmissometer, fluorometer, oxygen sensor, etc) along with discrete samples for salinity, dissolved oxygen (to 1 uM detection limits), plant pigments, redox tracers such as ammonium and nitrite, and dissolved nutrients at micro- and nanomolar levels; (4) ensure that proper QA/QC protocols are followed and reported, as well as fulfilling all GEOTRACES Intercalibration protocols; (5) prepare and deliver all hydrographic-type data to the GEOTRACES Data Center (and US data centers); and (6) coordinate cruise communications between all participating investigators, including preparation of a hydrographic report/publication.
Broader Impacts: The project is part of an international collaborative program that has forged strong partnerships in the intercalibration and implementation phases that are unprecedented in chemical oceanography. The science product of these collective missions will enhance our ability to understand how to interpret the chemical composition of the ocean, and interpret how climate change will affect ocean chemistry. Partnerships include contributions to the infrastructure of developing nations with overlapping interests in the study area, in this case Peru. There is a strong educational component to the program, with many Ph.D. students carrying out thesis research within the program.
Figure 1. The 2013 GEOTRACES EPZT Cruise Track. [click on the image to view a larger version]
U.S. GEOTRACES (U.S. GEOTRACES)
GEOTRACES is a SCOR sponsored program; and funding for program infrastructure development is provided by the U.S. National Science Foundation.
GEOTRACES gained momentum following a special symposium, S02: Biogeochemical cycling of trace elements and isotopes in the ocean and applications to constrain contemporary marine processes (GEOSECS II), at a 2003 Goldschmidt meeting convened in Japan. The GEOSECS II acronym referred to the Geochemical Ocean Section Studies To determine full water column distributions of selected trace elements and isotopes, including their concentration, chemical speciation, and physical form, along a sufficient number of sections in each ocean basin to establish the principal relationships between these distributions and with more traditional hydrographic parameters;
* To evaluate the sources, sinks, and internal cycling of these species and thereby characterize more completely the physical, chemical and biological processes regulating their distributions, and the sensitivity of these processes to global change; and
* To understand the processes that control the concentrations of geochemical species used for proxies of the past environment, both in the water column and in the substrates that reflect the water column.
GEOTRACES will be global in scope, consisting of ocean sections complemented by regional process studies. Sections and process studies will combine fieldwork, laboratory experiments and modelling. Beyond realizing the scientific objectives identified above, a natural outcome of this work will be to build a community of marine scientists who understand the processes regulating trace element cycles sufficiently well to exploit this knowledge reliably in future interdisciplinary studies.
Expand "Projects" below for information about and data resulting from individual US GEOTRACES research projects.
| Funding Source | Award |
|---|---|
| NSF Division of Ocean Sciences (NSF OCE) | |
| NSF Division of Ocean Sciences (NSF OCE) |
[ table of contents | back to top ]